Technical Details of Pharmaceutical Type I Glass Tubing
FIOLAX® and BORO-8330™ are ideally suited to pharmaceutical packaging thanks to their high levels of resistance to chemicals, temperature and pressure in combination with their excellent cosmetic and dimensional quality. All quality aspects are taylored to specific container formats such as syringes or vials. A variety of quality aspects can even be customized on demand.
Safe, strong and highly effective pharma packaging
General
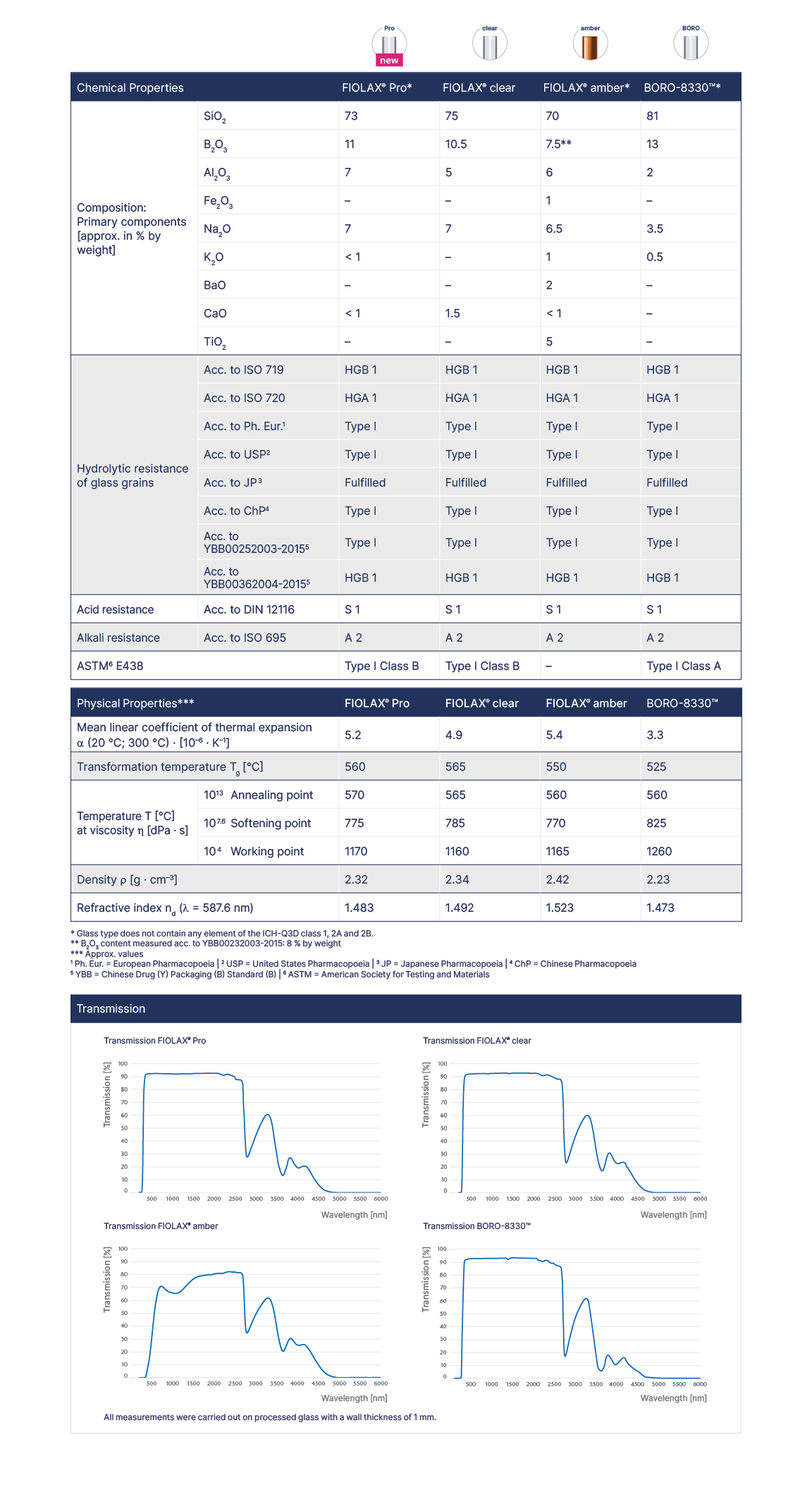
The technical specifications of FIOLAX® glass make it the world’s leading material for pharmaceutical Type I glass, with outstanding chemical resistance, neutrality, impermeability, and strength. FIOLAX® Pro and clear offer protective properties that will keep even the most sensitive of medicines stable and effective. FIOLAX® amber provides extra protection against ultraviolet rays and short-wave visible light. All our glass types meet all pharmaceutical requirements for manufacturing in accordance with GMP Guidelines.
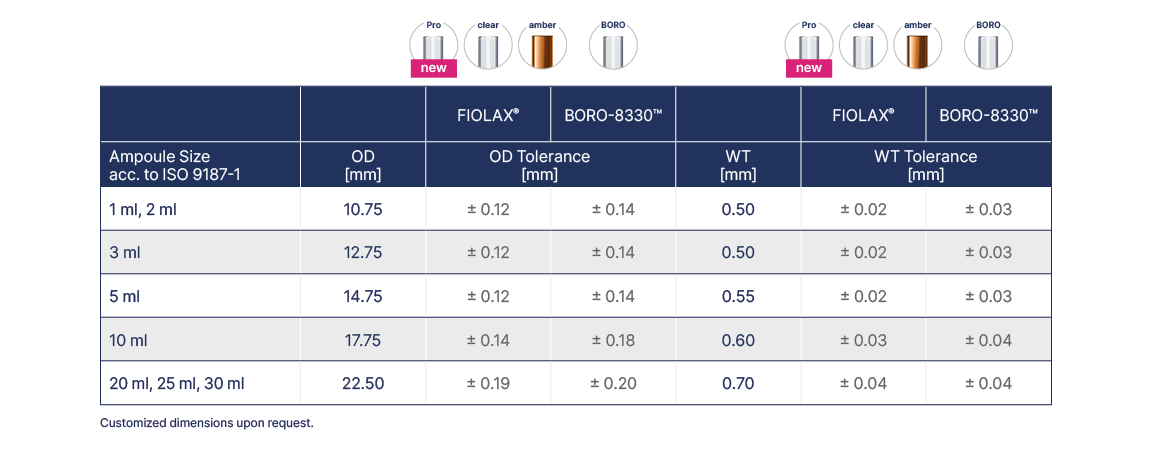
Ampoule Tubing
The excellent surface properties of FIOLAX® and BORO-8330™ provide permanent protection of the contents of ampoules, ensuring long term effectiveness of the packed pharmaceuticals. A 100% optical control of all FIOLAX® glass tubes involves the examination of every single tube for contamination or surface flaws, providing consistently excellent levels of quality for this high transparency glass.
The standard FIOLAX® tubing end finish for ampoules is DENSOCAN®, but other types and dimensions of tubing end finishes are available on request.
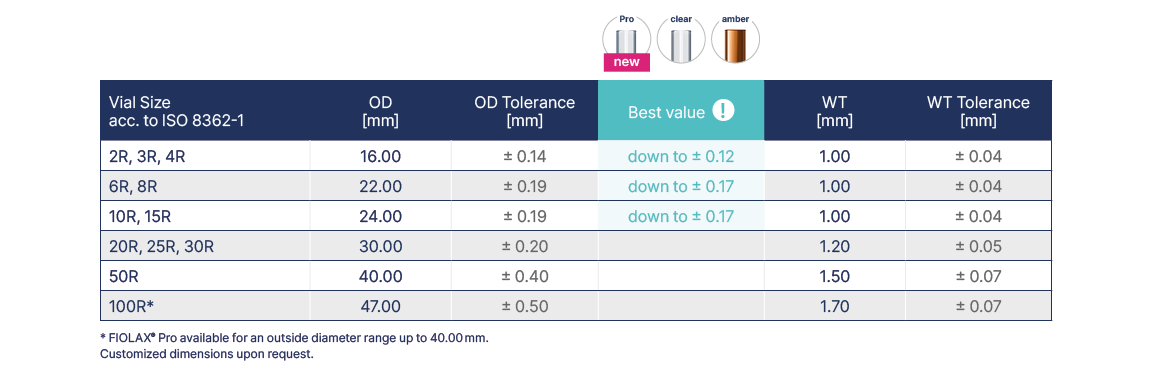
Vial Tubing
Whether the properties of medicines remain unchanged over long periods of time depends on the containers they are kept in. The outstanding chemical resistance, neutrality and impermeability of FIOLAX® and BORO-8330™ ensure an optimum protection of a vial’s contents against premature aging and loss of effectiveness. FIOLAX® amber additionally offers effective protection from ultra violet rays and short-wave visible light.
The standard tubing end finish for vials is DENSOCAN®, but other types and dimensions are available on request, such as a First Piece Version. Finally, all FIOLAX® glass tubes are subjected to 100% optical control throughout the entire production process.
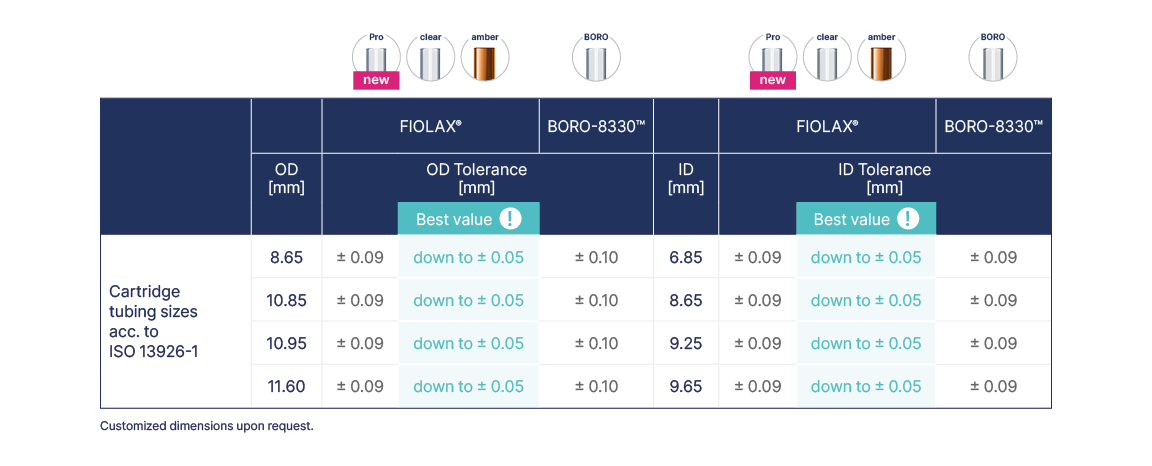
Cartridge Tubing
The narrow geometrical tolerances of FIOLAX® are not only advantageous for the processing and converting process, but also support the functionality of the cartridges that are frequently used in pen or pump systems. Overfill losses can be reduced and dosage accuracy is increased for the user, particularly in the case of multiple doses. Inside diameter tolerances down to ± 0.05 mm as a zero defect criteria can be produced upon request, depending on the chosen dimensions.
A 100% optical control integrated into the manufacturing process ensures the exceptional quality of FIOLAX®. The standard FIOLAX® tubing end finish for cartridges has both ends open. Other dimensions and types of tubing end are available on request.
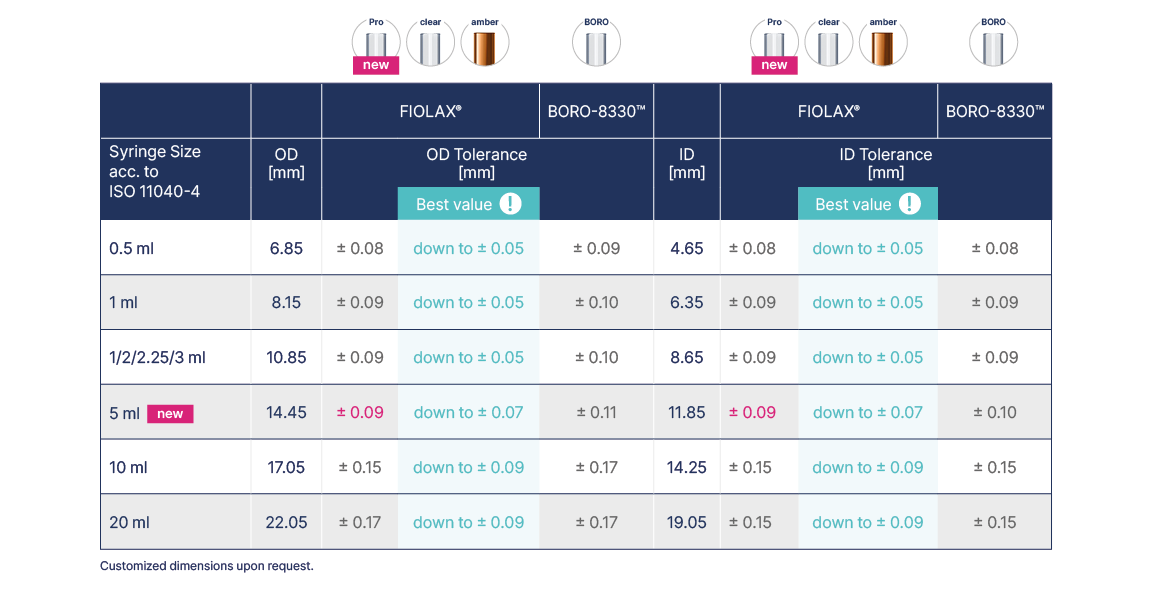
Syringe Tubing
Outperforming the ISO standard across the syringe tubing portfolio
The low geometric tolerances of FIOLAX® are vital in the production and processing of pre-fillable syringes. Already our standard FIOLAX® syringe tubing dimensions meet tighter OD and ID tolerances than required by ISO 11040-4. This supports our customers in enhancing the functionality of drug delivery systems, such as constant gliding forces and enhanced dosing accuracy.
The standard FIOLAX® tubing end finish for syringes has both ends open. In this manner, the open end can be used to form the first piece thus ensuring consistently high yields. Other dimensions and end finishes are also available on request.
To prevent breakage and scratches during transport, SCHOTT has developed DENSOPACK®, a packing method that, in combination with shrink wrapping, provides optimum protection for glass products in transit.

The DENSOCAN® process provides glass products with protection from contamination during transport and processing, as well as enhanced stability, lower particle load and improved converting process. Currently available for FIOLAX® clear, FIOLAX® amber and BORO-8330™.
Several customizing options are available for FIOLAX® clear, FIOLAX® amber and BORO-8330™ depending on customer requirements. One option is an anti-scratch coating, which protects the tubing during transport.

Dr. Haakon Wiedemann
Product Manager & Scientific Affairs Manager, SCHOTT Tubing Pharma