SCHOTT Vials A versatile portfolio to safely store injectable drugs
CONTACT USSecure storage of liquid drug formulations
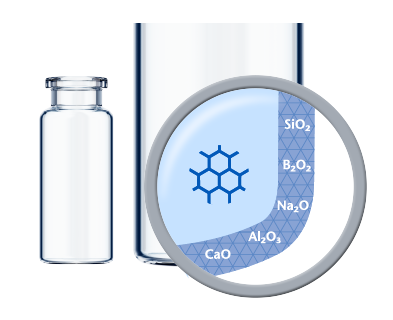
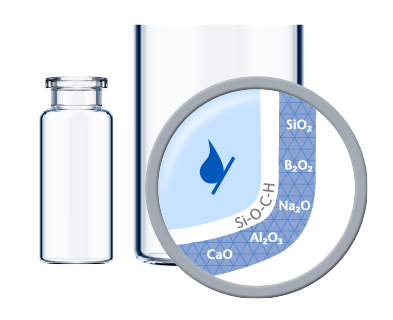
Made of Type I Borosilicate Glass, SCHOTT Pharma´s vials offer high chemical resistance for the secure storage of liquid drug formulations. Their accurate dimensions and superior cosmetic quality ensure an efficient fill-and-finish process and secure container closure integrity. In addition, special features such as improved inner surfaces, tighter geometries, and the option of inner and outer coatings fulfill further requirements of specific applications.High quality pharmaceutical vials with tight geometries
Our vials are available in two quality levels: StandardLine and TopLine. Thanks to 100% camera inspection, both offer tight geometries, but are differentiated by their cosmetic quality.Ensure shelf life stability for sensitive formulations
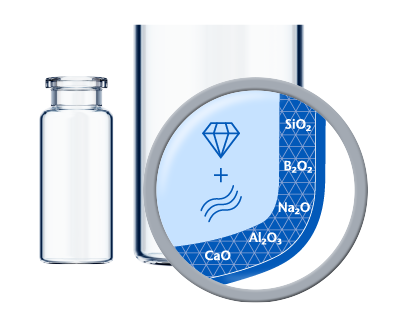
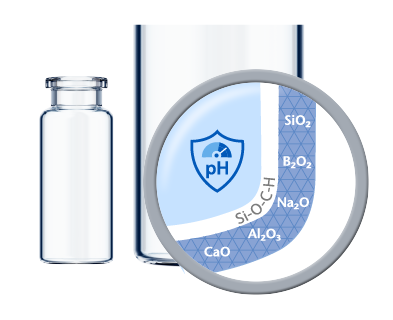
Based on the sensitivity of a formulation and additional processing steps such as terminal sterilization, a dedicated inner surface is often necessary to minimize drug-container interaction. EVERIC® pure offers an improved inner surface to fully control delamination and minimize leachables
Improve the efficiency of your fill-and-finish process
In order to reliably manufacture life-saving medicines, processes need to be stable and efficient. To improve the efficiency of fill-and-finish lines, packaging solutions such as SCHOTT TopLyo® vials can make a significant contribution.